fouling → obrastanje
1. Fouling is the deposition of insoluble materials, such as bacteria, colloids, oxides and water-borne debris, onto the surface of a reverse osmosis or ultrafiltration membrane. Fouling is associated with decreased flux rates and may also reduce the rejection rates of reverse osmosis membranes. Fouling also refers to the accumulation of normally inorganic deposits on heat exchanger tubing.
2. Fouling is an accumulation of marine organism deposits on a submerged metal surface.
Glauber’s salt → Glauberova sol
Glauber’s salt is sodium sulphate decahydrate (Na2SO4·10H2O). Loses water of hydration at 100 °C. Energy storage capacity is more than seven times that of water.
carbon → ugljik
Carbon has been known since ancient times. The origin of the name comes from the Latin word carbo meaning charcoal. Graphite form of carbon is a black, odourless, slippery solid. Graphite sublimes at 3825 °C. Diamond form is a clear or colored; an extremely hard solid. C60 is Buckminsterfullerine. Carbon black burns readily with oxidants. Carbon is made by burning organic compounds with insufficient oxygen. There are close to ten million known carbon compounds, many thousands of which are vital to organic and life processes. Radiocarbon dating uses the carbon-14 isotope to date old objects.
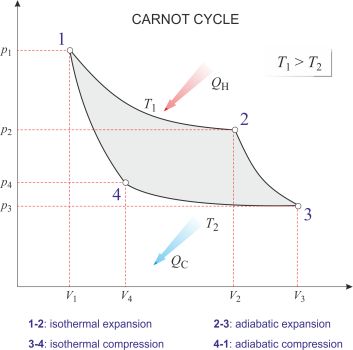
Carnot cycle → Carnotov kružni proces
Carnot cycle is the most efficient cycle of operations for a reversible heat engine. Published in 1824 by French physicist Nicolas Léonard Sadi Carnot (1796-1832), it consists of four operations on the working substance in the engine:
1-2: Isothermal expansion at thermodynamic temperature T1 with heat QH taken in.
2-3: Adiabatic expansion with a fall of temperature to T2.
3-4: Isothermal compression at temperature T2 with heat QC given out.
4-1: Adiabatic compression at temperature back to T1.
According to the Carnot principle, the efficiency of any reversible heat engine depends only on the temperature range through which it works, rather than the properties of the working substances.
convection → konvekcija
Convection is the process by which heat is transferred from one part of a fluid to another by movement of the fluid itself. There are two methods by which this can be carried out.
Natural convection, in which movement occurs as a result of gravity. Heat transferred through a fluid medium, such as air or water, by currents that result from the rising of less dense, warm fluid and the sinking of heavier, cooler fluid.
Forced convection is where hot fluid is transferred from one region to another by a mechanical means (fans or pumps).
copper → bakar
Copper has been known since ancient times. The origin of the name comes from the Latin word cuprum meaning the island of Cyprus famed for its copper mines. It is malleable, ductile, reddish-brown metal. Resistant to air and water. Exposed surfaces form greenish carbonate film. Pure copper occurs rarely in nature. Usually found in sulfides as in chalcopyrite (CuFeS2), coveline (CuS), chalcosine (Cu2S) or oxides like cuprite (Cu2O). Most often used as an electrical conductor. Also used in the manufacture of water pipes. Its alloys are used in jewellery and for coins.
cross-linking → umrežavanje
Cross-linking is an attachment of two chains of polymer molecules by bridges, composed of either an element, a group, or a compound, that join certain carbon atoms of the chains by primary chemical bonds, as indicated in the schematic diagram
Cross-linking occurs in nature in substances made up of polypeptide chains that are joined by the disulfide bonds of the cysteine residue, as in keratins or insulin. Cross-linking can be artificially effected, either adding a chemical substance (cross-linking agent), or by subjecting the polymer to high-energy radiation. Examples are: vulcanisation of rubber with sulphur, cross-linking of polystyrene with divinylbenzene, or cross-linking of polyethylene by means of high-energy radiation.
Cross-linking has the effect of changing a plastic from thermoplastic to thermosetting. Thus, it also increases strength, heat and electrical resistance, and especially resistance to solvents and other chemicals.
Curie → Curie
Maria Sklodowska-Curie (1867-1934) Polish-born French chemist who went to Paris in 1891. She married the physicist Pierre Curie (1859-1906) in 1985 and soon began work on seeking radioactive elements other than uranium in pitchblende (to account for its unexpectedly high radioactivity). By 1898 she had discovered radium and polonium although it took her years to purify them. In 1903 the Curies shared the Nobel Prize for physics with Henri Becquerel, who had discovered radioactivity.
desiccant dryers → sredstva za sušenje
Desiccant dryers (air dryers) use nonconsumable chemicals such as silica gel or active alumina, and can remove almost all the moisture from compressed air by adsorbing moisture on its surface. The method of regeneration, the process of removing adsorbed water from the desiccant, is the primary distinguishing feature among the various types of desiccant dryers. There are three ways to regenerate a desiccant: with air, internal or external heaters, or a heat pump.
Citing this page:
Generalic, Eni. "Toplina sublimacije." Croatian-English Chemistry Dictionary & Glossary. 29 June 2022. KTF-Split. {Date of access}. <https://glossary.periodni.com>.
Glossary
Periodic Table