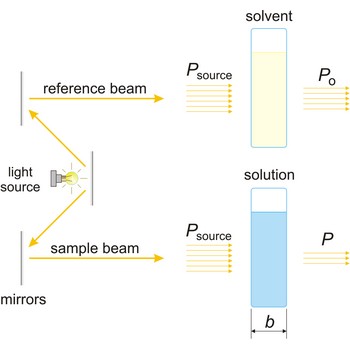
Beer’s law → Beerov zakon
Beer’s law (or Beer-Lambert law) is the functional relationship between the quantity measured in an absorption method (A) and the quantity sought, the analyte concentration (c). As a consequence of interactions between the photons and absorbing particles, the power of the beam is attenuated from Po to P. Beer’s law can be written
where A is the absorbance at a given wavelength of light, ε is the molar absorbtivity or extinction coefficient (L mol-1 cm-1), unique to each molecule and varying with wavelength, b is the length of light path through the sample (cm), and c is the concentration of the compound in solution (mol L-1).
berkelium → berkelij
Berkelium was discovered by Stanley G. Thompson, Albert Ghiorso and Glenn T. Seaborg (USA) in 1949. Named after Berkeley, a city in California, home of the University of California, USA. It is synthetic radioactive metal. Berkelium was made by bombarding americium with alpha particles.
beta radiation → beta zračenje
Streams of beta particles are known as beta ray or beta radiation. Beta rays may cause skin burns and are harmful within the body. A thin sheet of metal can afford protection to the skin.
Boltzmann constant → Boltzmannova konstanta
The Boltzmann constant (k or kB) is the physical constant describing the relationship between the thermodynamic temperature and the average kinetic energy of particles in a gas. It equals the molar gas constant R divided by the Avogadro constant NA and has the value 1.380 648 52(79)×10-23 J/K. It is named after the Austrian physicist Ludwig Eduard Boltzmann (1844-1906).
Brownian motion → Braunovo gibanje
Brownian motion is the continuous random movement of small particles suspended in a fluid, which arise from collisions with the fluid molecules. First observed by the British botanist R. Brown (1773-1858) when studying pollen particles. The effect is also visible in particles of smoke suspended in a gas.
californium → kalifornij
Californium was discovered by Stanley G. Thompson, Kenneth Street Jr. and Albert Ghiorso (USA) in 1950. Named after the State and University of California, USA. It is synthetic radioactive metal. Powerful neutron emitter. Californium was made by bombarding curium with helium ions.
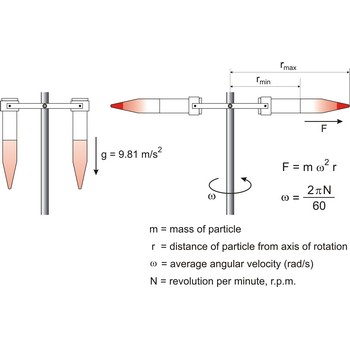
centrifugation → centrifugiranje
Centrifugation is sedimentation of particles under the influence of the centrifugal force and it is used for separation of superfine suspensions. At centrifuging forces up to 10 000 times greater than gravity force are used, and at ultracentrifuge up to 600 000 times as great.
coagulation → koaguliranje
Coagulation is a process of colloid particles merging into bigger ones. By removing the charge form a colloid ion, by increasing the temperature or by increasing electrolyte concentration, colloid particles will gather into bigger groups precipitate will emerge. Precipitate that is formed in this way (coagulate) is amorphic and considerably polluted with adsorbed pollutants.
colligative properties → koligativna svojstva
Colligative properties are properties which affect a solvent based on the number of molecules of solute present such as melting point, boiling point and osmotic pressure.
conjugated acid → konjugirana kiselina
Conjugated acid is a particle that develops after a base receives a proton.
Citing this page:
Generalic, Eni. "Subatomske čestice." Croatian-English Chemistry Dictionary & Glossary. 29 June 2022. KTF-Split. {Date of access}. <https://glossary.periodni.com>.
Glossary
Periodic Table