Helmholz free energy → Helmholzova slobodna energija
Helmholz free energy (A) is a thermodynamic function defined by A = U - TS, where U is the internal energy, S the entropy, and T the thermodynamic temperature. For a reversible isothermal process ΔA represents the useful work available.
hydrophilic → hidrofilan
Hydrophilic is having a strong tendency to bind or absorb water, which results in swelling and formation of reversible gels. This property is characteristic of carbohydrates.
intermediate → intermedijer
Intermediate is a molecular or ionic species that is formed (directly or indirectly) from the reactants and reacts further (directly or indirectly) to form the products of the reaction. It does not accumulate during the course of the reaction.
coenzyme a → koenzim a
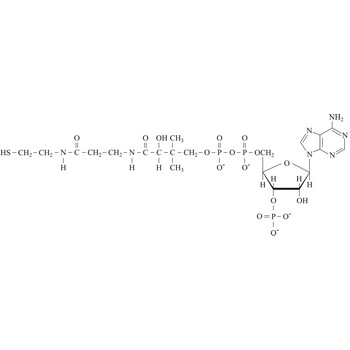
Coenzyme A (CoA) is an essential metabolic cofactor synthesized from cysteine, pantothenate (vitamin B5), and ATP. CoA plays important roles in many metabolic pathways, including the tricarboxylic acid (TCA) cycle, and the synthesis and oxidation of fatty acids. One of the main functions of CoA is the carrying and transfer of acyl groups. Acylated derivatives (acetyl-CoA) are critical intermediates in many metabolic reactions.
collision theory → teorija sudara
Collision theory is theory that explains how chemical reactions take place and why rates of reaction alter. For a reaction to occur the reactant particles must collide. Only a certain fraction of the total collisions cause chemical change; these are called successful collisions. The successful collisions have sufficient energy (activation energy) at the moment of impact to break the existing bonds and form new bonds, resulting in the products of the reaction. Increasing the concentration of the reactants and raising the temperature bring about more collisions and therefore more successful collisions, increasing the rate of reaction.
colloid ion → koloidni ion
Colloid ions emerge when colloid particles adsorb certain type of ion from solution and thus become charged with the same charge. The charge can also originate form a chemical reaction of colloid particle’s surface. Colloid ions formed by absorption of silver chloride particle can be show as follows:
Adsorbed layer is monomolecular (one molecule thick) and which type of ion will be formed depends upon which ions are present in a greater number in the solution in. Because of this colloid particles are charged with the same charge, mutual repelling occurs, and the colloid solution becomes stable. Colloid charge can be determined by electrophoresis.
irreversible galvanic cell → nepovrativi galvanski članak
Irreversible galvanic cell is a chemical source of direct current, in which reactions that take place on the electrodes are irreversible.
Citing this page:
Generalic, Eni. "Reverzibilna reakcija." Croatian-English Chemistry Dictionary & Glossary. 29 June 2022. KTF-Split. {Date of access}. <https://glossary.periodni.com>.
Glossary
Periodic Table