joule → džul
Joule (J) is the SI derived unit of energy, work, and heat. The joule is the work done when the point of application of a force of one newton is displaced a distance of one metre in the direction of the force (J = N m). The unit was named after the British scientist James Prescott Joule (1818-1889).
meitnerium → meitnerij
Meitnerium was discovered by Peter Armbruster, Gottfried Münzenber and their co-workers at the Heavy Ion Research Laboratory (Gesellschaft für Schwerionenforschung, GSI) in Darmstadt, Germany in 1982. Named in honour of Lise Meitner (1878-1968), the Austrian physicist. It is synthetic radioactive metal. Meitnerium was produced bybombarding bismuth-209 with iron-58.
gas washing bottle → boca ispiralica
Gas washing bottle or Drechsel bottle provide an inexpensive but effective method for washing or drying gases. The gas enters the bottle through the top of the central vertical tube, the lower end of which is below the surface of the washing medium. To maximize surface area contact of the gas to the liquid, a gas stream is slowly blown into the vessel through the fritted glass tip so that it breaks up the gas into many tiny bubbles. After bubbling through the medium, the gas rises to the top and exits through the side tube. It is named after the German chemist Edmund Drechsel (1843-1897).
glucose → glukoza
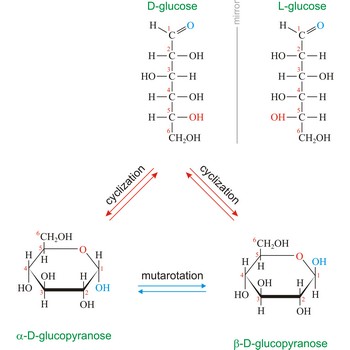
Glucose (grape sugar, blood sugar), C6H12O6, is an aldohexose (a monosaccharide sugar having six carbon atoms and an aldehyde group). An older common name for glucose is dextrose, after its dextrorotatory property of rotating plane polarized light to the right. Glucose in free (in sweet fruits and honey) or combined form (sucrose, starch, cellulose, glycogen) is is probably the most abundant organic compound in nature. During the photosynthesis process, plants use energy from the sun, water from the soil and carbon dioxide gas from the air to make glucose. In cellular respiration, glucose is ultimately broken down to yield carbon dioxide and water, and the energy from this process is stored as ATP molecules (36 molecules of ATP across all processes).
Naturally occurring glucose is D isomers (OH group on the stereogenic carbon farthest from the aldehyde group, C-5, is to the right in the Fischer projection). Although often displayed as an open chain structure, glucose and most common sugars exist as ring structures. In the α form, the hydroxyl group attached to C-1 and the CH2OH attached to C-5 are located on opposite sides of the ring. β-glucose has these two groups on the same side of the ring. The full names for these two anomers of glucose are α-D-glucopyranose and β-D-glucopyranose.
mendelevium → mendelevij
Mendelevium was discovered by Albert Ghiorso, Bernard G. Harvey, Gregory R. Choppin, Stanley G. Thompson and Glenn T. Seaborg (USA) in 1955. Named in honour of Dimitri Mendeljejev, the Russian chemist who devised the periodic table. It is synthetic radioactive metal. Mendelevium was made by bombarding einsteinium with helium ions.
neptunium → neptunij
Neptunium was discovered by Edwin M. McMillan and P. H. Abelson (USA) in 1940. Named after the planet Neptune. It is rare, silvery radioactive metal. Resists alkalis; reacts with oxygen and acids. Attacked by steam. Radiotoxic. Neptunium was produced by bombarding uranium with slow neutrons.
glutamine → glutamin
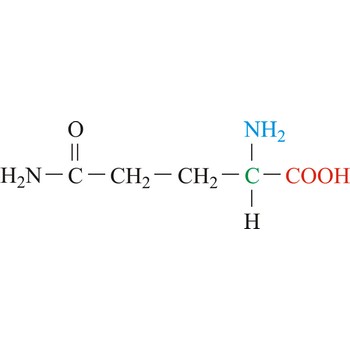
Glutamine is neutral amino acids with polar side chains. It serves as an important carrier of ammonia and contributes it to the formation of urea and purines. Glutamine is not recognized as an essential amino acid but may become conditionally essential in certain situations, including intensive athletic training or certain gastrointestinal disorders. It is synthesized by the enzyme glutamine synthetase from glutamate and ammonia.
- Abbreviations: Gln, Q
- IUPAC name: 2,5-diamino-5-oxopentanoic acid
- Molecular formula: C5H10N2O3
- Molecular weight: 146.14 g/mol
glycogen → glikogen
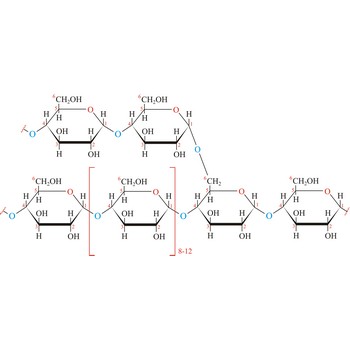
Glycogen (animal starch) is a polysaccharide that serves the same energy storage function in animals that starch serves in plants. Dietary carbohydrates not needed for immediate energy are converted by the body to glycogen for long term storage (principally in muscle and liver cells). Like amylopectin found in starch, glycogen is a polymer of α(1→4)-linked subunits of glucose, with α(1→6)-linked branches. Glycogen molecules are larger than those of amylopectin (up to 100 000 glucose units) and contain even more branches. Branch points occur about every 10 residues in glycogen and about every 25 residues in amylopectin. The branching also creates lots of ends for enzyme attack and provides for rapid release of glucose when it is needed.
groups in periodic system of elements → skupine periodnog sustava
Periodic system of elements is divided into 18 groups of chemical elements. Elements belonging to the same group have a same number of valence electrons and similar chemical properties. Elements of main groups are in 1., 2., and in groups 13. to 18. Different groups of elements can be named according to the first element in the group (elements of boron group, elements of carbon group), or they have some special names (noble gases, halogenic elements, halyde elements, earthalkali and alkali metals).
Nessler’s reagent → Nesslerov reagens
Nessler’s reagent is a solution of mercury(II) iodide (HgI2) in potassium iodide and potassium hydroxide named after the German chemist Julius Nessler (1827-1905). It is used in testing for ammonia, with which it forms a brown coloration or precipitate.
Citing this page:
Generalic, Eni. "Nginx server_name regular expression." Croatian-English Chemistry Dictionary & Glossary. 29 June 2022. KTF-Split. {Date of access}. <https://glossary.periodni.com>.
Glossary
Periodic Table