transference number → prijenosni broj
Transference number of an ion is the fraction of the total current that is carried by that ion during electrolysis. Different ions carry different fractions of the current because different ions move at different speeds under the same potential gradient. In general, a cation and an anion differ in the amount of current they can carry during electrolysis.
glycogen → glikogen
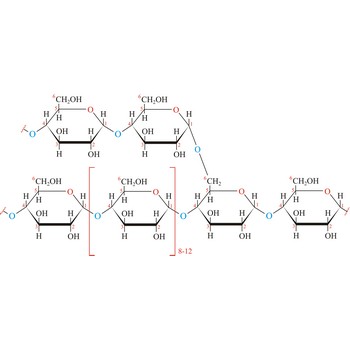
Glycogen (animal starch) is a polysaccharide that serves the same energy storage function in animals that starch serves in plants. Dietary carbohydrates not needed for immediate energy are converted by the body to glycogen for long term storage (principally in muscle and liver cells). Like amylopectin found in starch, glycogen is a polymer of α(1→4)-linked subunits of glucose, with α(1→6)-linked branches. Glycogen molecules are larger than those of amylopectin (up to 100 000 glucose units) and contain even more branches. Branch points occur about every 10 residues in glycogen and about every 25 residues in amylopectin. The branching also creates lots of ends for enzyme attack and provides for rapid release of glucose when it is needed.
lead-acid battery → olovni akumulator
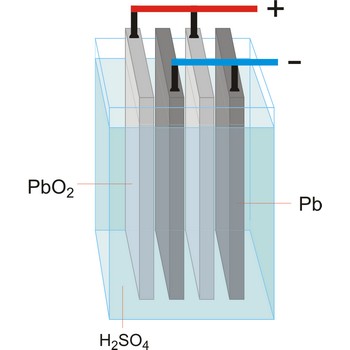
Lead-acid battery is a electrical storage device that uses a reversible chemical reaction to store energy. It was invented in 1859 by French physicist Gaston Planté. Lead-acid batteries are composed of a lead(IV) oxide cathode, a sponge metallic lead anode and a sulphuric acid solution electrolyte.
In charging, the electrical energy supplied to the battery is changed to chemical energy and stored. The chemical reaction during recharge is normally written:
In discharging, the chemical energy stored in the battery is changed to electrical energy. During discharge, lead sulfate (PbSO4) is formed on both the positive and negative plates. The chemical reaction during discharge is normally written:
Lead acid batteries are low cost, robust, tolerant to abuse, tried and tested. For higher power applications with intermittent loads however, they are generally too big and heavy and they suffer from a shorter cycle life.
voltametry → voltametrija
Voltametry is a common name for a large group of instrumental techniques which are based on measuring the electric current formed by a continuous potential shifting on the electrodes.
piezoelectric effect → piezoelektrični efekt
Piezoelectric effect is voltage produced between surfaces of a solid dielectric (nonconducting substance) when a mechanical stress is applied to it. A small current may be produced as well. The effect, discovered by the French physicist Pierre Curie (1859-1906) in 1883, is exhibited by certain crystals, e.g., quartz and Rochelle salt, and ceramic materials. When a voltage is applied across certain surfaces of a solid that exhibits the piezoelectric effect, the solid undergoes a mechanical distortion. Piezoelectric materials are used in transducers, e.g., phonograph cartridges, microphones, and strain gauges, which produce an electrical output from a mechanical input, and in earphones and ultrasonic radiators, which produce a mechanical output from an electrical input.
Volta, Alessandro → Volta, Alessandro
The Italian physicist Alessandro Giuseppe Antonio Anastasio Volta (1745-1827) was the inventor of the voltaic pile, the first electric battery (1800). In 1775 he invented the electrophorus, a device that, once electrically charged by having been rubbed, could transfer charge to other objects. Between 1776 and 1778, Volta discovered and isolated methane gas (CH4). The electrical unit known as the volt was named in his honor.
voltaic pile → Voltin stup
Voltaic pile was the first device that produced a continuous electric current. The first piles constructed by the Italian physicist Alessandro Volta (1745-1827) in 1800 comprised alternating silver and zinc discs separated by cardboard soaked in brine. The pile can be stacked as high as you like, and each layer will increase the voltage by a fixed amount.
X-ray tube → rendgenska cijev
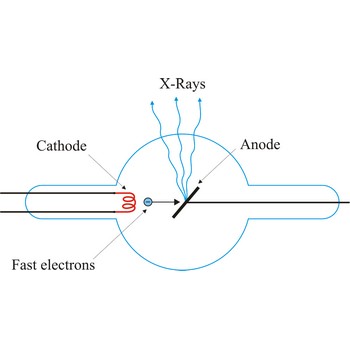
X-ray tube is a cathode ray tube that focuses energetic streams of electrons on a metal target, causing the metal to emit X-rays. The basic principle of the X-ray tube has not changed significantly since Roentgen's 1895 discovery. Current applied to a metal cathode (about 50 000 V) produces free electrons. The X-rays are produced when the rapidly moving electrons are suddenly stopped as they strike the metal target of the tube.
Citing this page:
Generalic, Eni. "Napon razlaganja." Croatian-English Chemistry Dictionary & Glossary. 29 June 2022. KTF-Split. {Date of access}. <https://glossary.periodni.com>.
Glossary
Periodic Table