law of chemical equilibrium → zakon o kemijskoj ravnoteži
Law of chemical equilibrium (also called the law of mass action) states that the rate at which a substance reacts is proportional to its active mass (i.e. to its molar concentration). Thus, the velocity of a chemical reaction is proportional to the product of the concentration of the reactants.
hexagonal lattice → heksagonska rešetka
Hexagonal lattice has lattice points at the twelve corners of the hexagonal prism and at the centers of the two hexagonal faces of the unit cell. It has unit cell vectors a=b≠c and interaxial angles α=β=90° and γ=120°.
hydrolysis → hidroliza
Hydrolysis is a chemical reaction in which water reacts with another substance to form two or more new substances. This involves ionisation of the water molecule, as well as splitting of the compound hydrolysed, e.g.
Examples are conversion of starch to glucose by water in the presence of suitable catalysts and a reaction of the ions of a dissolved salt to form various products, such as acids, complex ions, etc.
ideal solution → idealna otopina
Ideal solution is a solution in which solvent-solvent and solvent-solute interactions are identical, so that properties such as volume and enthalpy are exactly additive. Ideal solutions follow Raoult’s law, which states that the vapour pressure pi of component i is pi = xi pi*, where xi is the mole fraction of component i and pi* the vapour pressure of the pure substance i.
indicator → indikator
Indicator is a substance used to show the presence of a chemical substance or ion by its colour. Acid-base indicators are compounds, such as phenolphtaleine and methyl orange, which change colour reversibly, depending on whether the solution is acidic or basic. Oxidation-reduction indicators are substances that show a reversible colour change between oxidised and reduced forms.
levorotatory → lijevokretan
Levorotatory is refers to an optically active substance that rotates the plane of plane polarised light counterclockwise.
Lewis number → Lewisova značajka
Lewis number (Le) is a dimensionless quantity used in fluid mechanics, defined by
where a is thermal diffusivity and D is diffusion coefficient.
magnetochemistry → magnetokemija
Magnetochemistry is a branch of physical chemistry which studies relations between magnetism and the chemical structure of matter.
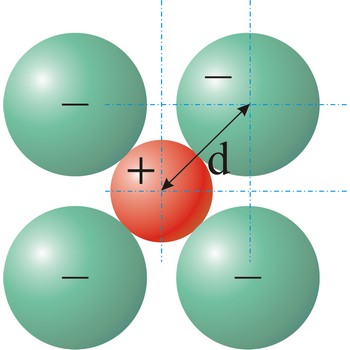
ionic radius → ionski radijus
Ionic radius is the radius of anions and cations in crystalline ionic compounds, as determined by consistently partitioning the center-to-center distance of ions in those compounds. In general, negative ions have larger ionic radii than positive ions.
ionisation → ionizacija
Ionisation is the process of producing ions. Certain molecules ionise in a solution; for example, acids ionise when dissolved in water.
Electron transfer also causes ionisation in certain reactions, for example sodium and chlorine react by transfer of a valence electron from the sodium atom to the chlorine atom to form the ions that constitute a sodium chloride crystal.
Citing this page:
Generalic, Eni. "Kristalna tvar." Croatian-English Chemistry Dictionary & Glossary. 29 June 2022. KTF-Split. {Date of access}. <https://glossary.periodni.com>.
Glossary
Periodic Table