magnetochemistry → magnetokemija
Magnetochemistry is a branch of physical chemistry which studies relations between magnetism and the chemical structure of matter.
metabolism → metabolizam
Metabolism is a sum of all chemical and physiological processes by which the body builds and maintains itself. It is a process of building the body’s molecular structures from nutrients (anabolism) and breaking them down for energy (catabolism).
electrogravimetry → electrogravimetrija
Electrogravimetry is an electroanalytical technique in which the substance to be determined (usually a metal) is deposited out on an electrode which is weighed before and after the experiment. The potential of the electrode must be carefully chosen to ensure that only the metal do be determined will deposit.
mineral acid → mineralna kiselina
Mineral acid is an acid made from minerals by chemical reaction, e.g. hydrochloric acid is produced from sodium chloride and sulphuric acid is made from sulphur.
nascent state → nascentno stanje
Nascent state is an especially active state of an element in a moment when it is released from a compound during chemical reaction, e.g. nascent hydrogen.
negative pole → negativan pol
Negative pole is that half-cell in electrochemical cell that has the most negative electrode potential.
neon → neon
Neon was discovered by Sir William Ramsay and Morris W. Travers (England) in 1898. The origin of the name comes from the Greek word neon meaning new. It is colourless, odourless inert gas. Non-reactive even with fluorine. Fourth most abundant element in the universe. Neon is obtained by liquefaction of air and separated from the other gases by fractional distillation. Primarily for lighting.
overpotential → prenapon
Overpotential (η) is a potential that must be applied in an electrolytic cell in addition to the theoretical potential required to liberate a given substance at an electrode. The value depends on the electrode material and on the current density.
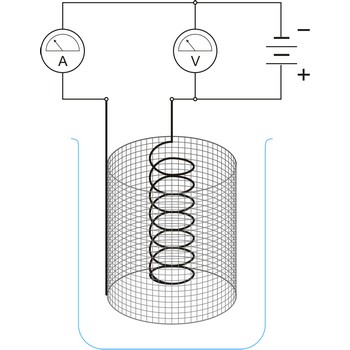
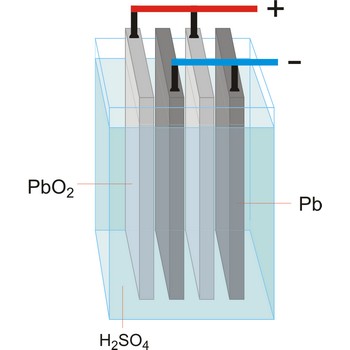
electrolytic cell → elektrolitska ćelija
Electrolytic cell is an electrochemical cell that converts electrical energy into chemical energy. The chemical reactions do not occur spontaneously at the electrodes when they are connected through an external circuit. The reaction must be forced by applying an external electric current. It is used to store electrical energy in chemical form (rechargeable battery). It is also used to decompose or produce (synthesise) new chemicals by the application of electrical power. This process is called electrolysis, e.g., water can be decomposed into hydrogen gas and oxygen gas. The free energy change of the overall cell reaction is positive.
Citing this page:
Generalic, Eni. "Kemijski potencijal." Croatian-English Chemistry Dictionary & Glossary. 29 June 2022. KTF-Split. {Date of access}. <https://glossary.periodni.com>.
Glossary
Periodic Table