halocarbon → halogenirani ugljikovodik
Halocarbon is a compound containing no elements other than carbon, one or more halogens, and sometimes hydrogen. The simplest are compounds such as tetrachloromethane (CCl4), tetrabromomethane (CBr4), etc. The lower members of the various homologous series are used as refrigerants, propellant gases, fireextinguishing agents, and blowing agents for urethane foams. When polymerized, they yield plastics characterized by extreme chemical resistance, high electrical resistivity, and good heat resistance.
helium → helij
Helium was discovered by Pierre Jules César Janssen (France) and Sir William Ramsay (Scotland) in 1868. The origin of the name comes from the Greek word helios meaning sun. It is light, odourless, colourless inert gas. Second most abundant element in the universe. Helium is found in natural gas deposits from wells in Texas, Oklahoma and Kansas. Used in balloons, deep sea diving and welding. Also used in very low temperature research.
Hooke’s law → Hookeov zakon
Hooke’s law stating that the deformation of a body is proportional to the magnitude of the deforming force, provided that the body’s elastic limit (see elasticity) is not exceeded. If the elastic limit is not reached, the body will return to its original size once the force is removed. The law was discovered by English physicist Robert Hooke in 1676.
If a body on elastic spring is displaced from its equilibrium position (i.e. if the spring is stretched or compressed), a restitution force tries to return the body back in its equilibrium position. The magnitude of that force is proportional to the displacement of the body
Where F is the restitutional (elastic) force, x is the displacement of the body and k is the spring constant, which depends on dimensions, shape and material of the spring.
hydrolysis → hidroliza
Hydrolysis is a chemical reaction in which water reacts with another substance to form two or more new substances. This involves ionisation of the water molecule, as well as splitting of the compound hydrolysed, e.g.
Examples are conversion of starch to glucose by water in the presence of suitable catalysts and a reaction of the ions of a dissolved salt to form various products, such as acids, complex ions, etc.
indicator → indikator
Indicator is a substance used to show the presence of a chemical substance or ion by its colour. Acid-base indicators are compounds, such as phenolphtaleine and methyl orange, which change colour reversibly, depending on whether the solution is acidic or basic. Oxidation-reduction indicators are substances that show a reversible colour change between oxidised and reduced forms.
ion-product constant → ionski produkt vode
The ion-product constant. For the reaction:
the equilibrium expression would be:
Note that all pure liquid terms are omitted, hence H2O does not appear in the denominator. At 25 °C
krypton → kripton
Krypton was discovered by Sir William Ramsay and Morris W. Travers (England) in 1898. The origin of the name comes from the Greek word kryptos meaning hidden. It is colourless, odourless rare noble gas. Reacts only with fluorine. Krypton is obtained from production of liquid air. Used in lighting products. Some is used as inert filler-gas in incandescent bulbs. Some is mixed with argon in fluorescent lamps. The most important use is in flashing stroboscopic lamps that outline airport runways.
lactose → laktoza
Lactose (milk sugar) is a disaccharide comprising one glucose molecule linked to a galactose molecule by an β(1→4)-glycosidic linkage. Lactose has a beta acetal. Lactose is manufactured by the mammary gland and occurs only in milk (from 4 % to 7 %). Lactose intolerance is a common medical condition that results in diarrhea, abdominal pain, and flatulence and is caused by reduced or absent activity of enzyme lactase.
Like cellobiose and maltose, lactose is a reducing sugar. All reducing sugar undergo mutarotation in aqueous solution. The equilibrium mixture at 20 °C is composed of 62.7 % β-lactose (β-D-galactopyranosyl-(1→4)-β-D-glucopyranose) and 37.3 % α-lactose (β-D-galactopyranosyl-(1→4)-α-D-glucopyranose).
law of conservation of mass → zakon o očuvanju mase
Law of conservation of mass states that no detectable gain or loss in mass occurs in chemical reactions. The state of a substance may change in a chemical reaction, for example, from a solid to a gas, but its total mass will not change. Note that the energy released (exothermic) or adsorbed (endothermic) in a chemical reaction is a result of energy transfer between atoms and their environment.
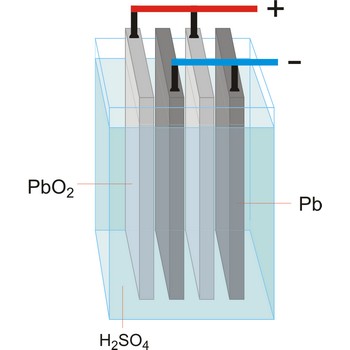
lead-acid battery → olovni akumulator
Lead-acid battery is a electrical storage device that uses a reversible chemical reaction to store energy. It was invented in 1859 by French physicist Gaston Planté. Lead-acid batteries are composed of a lead(IV) oxide cathode, a sponge metallic lead anode and a sulphuric acid solution electrolyte.
In charging, the electrical energy supplied to the battery is changed to chemical energy and stored. The chemical reaction during recharge is normally written:
In discharging, the chemical energy stored in the battery is changed to electrical energy. During discharge, lead sulfate (PbSO4) is formed on both the positive and negative plates. The chemical reaction during discharge is normally written:
Lead acid batteries are low cost, robust, tolerant to abuse, tried and tested. For higher power applications with intermittent loads however, they are generally too big and heavy and they suffer from a shorter cycle life.
Citing this page:
Generalic, Eni. "Kemijska ravnoteža." Croatian-English Chemistry Dictionary & Glossary. 29 June 2022. KTF-Split. {Date of access}. <https://glossary.periodni.com>.
Glossary
Periodic Table