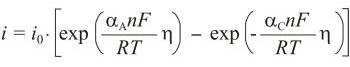chemical property → kemijsko svojstvo
Chemical property is a property observed when a substance undergoes a transformation into one or more new substances. Measurement of a chemical property involves a chemical change. For example, determining the flammability of petrol involves burning it, producing carbon dioxide and water.
Butler-Volmer equation → Butler-Volmerova jednadžba
Butler-Volmer equation is an activation controlled reaction, the one for which the rate of reaction is controlled solely by the rate of the electrochemical charge transfer process, which is in turn an activation-controlled process. This gives rise to kinetics that are described by the Butler-Volmer equation:
where io is exchange current density, η is overpotential (η = E - Eo), n is number of electrons, αA is anodic transfer coefficient, and αC is cathodic transfer coefficient
chemical technology → kemijska tehnologija
Chemical technology is a branch of applied chemistry that concerns technical methods and devices in order to manufacture a chemical product.
complete ionic equation → potpuna ionska jednadžba
Complete ionic equation is a balanced equation that describes a reaction occurring in a solution, in which all strong electrolytes are written as dissociated ions.
chemical change → kemijska promjena
Chemical change is a process which results in the production of one or more new materials. The system within which the process takes place is called a chemical system. A chemical change is also known as a chemical reaction, where one substance is converted into one or more different substances. When sodium and chlorine react to produce sodium chloride, a chemical reaction has taken place.
chemical compound formula → formula kemijskog spoja
Chemical elements are represented by their symbols, and chemical compounds are represented by a group of symbols of those elements from which the compound is composed. That group of symbols, which shows which atoms and in which number relation they are present in certain compound is called a chemical compound formula.
In a formula chemical symbols show which element is present in a certain compound, and its index shows how much of that element there is in a certain compound. From sulphuric acid formula H2SO4 we can see that one molecule of sulphuric acid consists of two atoms of hydrogen, one atom of sulphur and four atoms of oxygen.
chemical potential → kemijski potencijal
For a mixture of substances, the chemical potential of constituent B (μB) is defined as the partial derivative of the Gibbs energy G with respect to the amount (number of moles) of B, with temperature, pressure, and amounts of all other constituents held constant.
Also called partial molar Gibbs energy. Components are in equilibrium if their chemical potentials are equal.
chemical weapons → kemijsko oružje
The Chemical Weapons Convention, article 2, paragraph 1 defines chemical weapons thus:
Chemical weapons means the following, together or separately:
(a) Toxic chemicals and their precursors, except where intended for purposes not prohibited under this Convention, as long as the types and quantities are consistent with such purposes;
(b) Munitions and devices, specifically designed to cause death or other harm through the toxic properties of those toxic chemicals specified in subparagraph (a), which would be released as a result of the employment of such munitions and devices;
(c) Any equipment specifically designed for use directly in connection with the employment of munitions and devices specified in subparagraph (b).
Clapeyron equation → Clapeyronova jednadžba
Clapeyron equation (also called the Clausius-Clapeyron equation) is a relation between pressure and temperature of two phases of a pure substance that are in equilibrium,
where ΔtrsS is the difference in entropy between the phases and ΔtrsV the corresponding difference in volume.
equation of state → jednadžba stanja
Equation of state is an equation relating the pressure, volume, and temperature of a substance or system. Equation of state for ideal gas
where p is pressure, V molar volume, T temperature, and R the molar gas constant (8.314 JK-1mol-1).
Citing this page:
Generalic, Eni. "Izjednačavanje kemijske jednadžbe." Croatian-English Chemistry Dictionary & Glossary. 29 June 2022. KTF-Split. {Date of access}. <https://glossary.periodni.com>.
Glossary
Periodic Table