critical volume → kritični volumen
Critical volume is the volume of a fixed mass of a fluid at critical temperature and pressure.
critical pressure → kritični tlak
Critical pressure is the pressure of a fluid in its critical point; i.e. when it is at its critical temperature and critical volume.
absolute volume → apsolutni volumen
Absolute volume is the total volume of the particles in a granular material, including both permeable and impermeable voids but excluding spaces between particles.
critical mass → kritična masa
Critical mass is the minimum mass of a fissionable material (235U or 239Pu) that will initiate an uncontrolled chain reaction as in an atomic bomb. The critical mass of pure 239Pu is about 4.5 kg, and of 235U about 15 kg.
molar volume → molarni volumen
Molar volume is the volume occupied by substance per unit amount of substance. The volume of the gas at 0 °C and 101 325 Pa is 22.4 dm3mol-1.
volume ratio → volumenski omjer
Volume ratio is equal to volume (VA) of one component and volume (VB) of other component’s proportion.
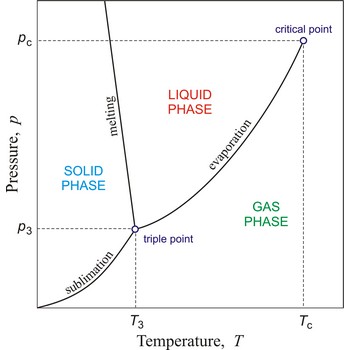
critical point → kritična točka
In general, critical point is the point on the phase diagram of a two-phase system at which the two coexisting phases have identical properties and therefore represent a single phase. At the liquid-gas critical point of a pure substance, the distinction between liquid and gas vanishes, and the vapour pressure curve ends. The coordinates of this point are called the critical temperature and critical pressure. Above the critical temperature it is not possible to liquefy the substance.
critical temperature → kritična temperatura
Critical temperature is the temperature of the liquid-vapour critical point, that is, the temperature above which a gas cannot be liquefied by an increase of pressure.
volume concentration → volumenska koncentracija
Volume concentration (σ) is equal to volume (VA) of solute and volume (V) of solution proportion. Volume concentration differs from volume fraction because the sum of solution components volume is almost always different than the solution volume.
Citing this page:
Generalic, Eni. "Critical volume." Croatian-English Chemistry Dictionary & Glossary. 29 June 2022. KTF-Split. {Date of access}. <https://glossary.periodni.com>.
Glossary
Periodic Table